TITANIUM(II) CHLORIDE
TITANIUM(II) CHLORIDE
CAS: 10049-06-6
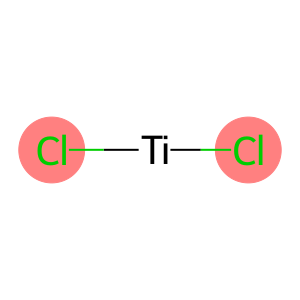
Molecular Formula: Cl2Ti
TITANIUM(II) CHLORIDE - Names and Identifiers
| Name | TITANIUM(II) CHLORIDE |
| Synonyms | TiCl2 titanium dichloride TITANIUM(II) CHLORIDE titaniumchloride(ticl2) Titanium(II) dichloride Titanium chloride (TiCl2) |
| CAS | 10049-06-6 |
| EINECS | 233-164-9 |
TITANIUM(II) CHLORIDE - Physico-chemical Properties
| Molecular Formula | Cl2Ti |
| Molar Mass | 118.77 |
| Density | 3.13 |
| Melting Point | 1035° |
| Boling Point | 1652.26°C (estimate) |
| Water Solubility | decomposed by H2O evolving H2and forming TiCl3 [KIR83]; soluble alcohol; insoluble chloroform, ether, CS2 [MER06] |
| Appearance | powder |
| Merck | 13,9548 |
| Stability | Stability Pyrophoric. Reacts violently with water. Incompatible with strong oxidizing agents. May decompose upon exposure to air. |
TITANIUM(II) CHLORIDE - Risk and Safety
| Risk Codes | R14/15 - R17 - Spontaneously flammable in air R34 - Causes burns |
| Safety Description | S16 - Keep away from sources of ignition. S24 - Avoid contact with skin. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3200 4.2/PG 1 |
| WGK Germany | 3 |
| Hazard Class | 4.2 |
| Packing Group | I |
TITANIUM(II) CHLORIDE - References
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| production method | in a rare gas atmosphere, 3g of titanium trichloride was placed in an eggplant-shaped flask made of borosilicate glass, the flask was connected to a liquid nitrogen cooling trap nozzle and then to a vacuum pump. When the reaction temperature is higher than 475 ° C., since the generated titanium dichloride undergoes a disproportionation reaction to produce titanium and titanium tetrachloride, it should be noted that the reaction temperature should not be too high. Since a small amount of titanium trichloride is partially vaporized and precipitated at the bottleneck during heating, the precipitate can be often vibrated and returned to the heating section. After the completion of the reaction, a melting seal was performed at a necking prepared in advance in the upper portion of the flask. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: TITANIUM(II) CHLORIDE Request for quotation
CAS: 10049-06-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 10049-06-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: TITANIUM(II) CHLORIDE Request for quotation
CAS: 10049-06-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 10049-06-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History
